Lead-acid battery internal resistance - QuickField simulation example
Lead-acid battery consists of two lead grid plates submerged into the electrolyte - concentrated aqueous sulfuric acid solution. Positive plate is covered by the lead dioxide which is formed by the chemical reaction during the charge process, and second grid plate is a negative electrode.
How to find lead acid battery internal resistance?
Engineering answer Typical applications Geometry
Given
Task
Solution
Results
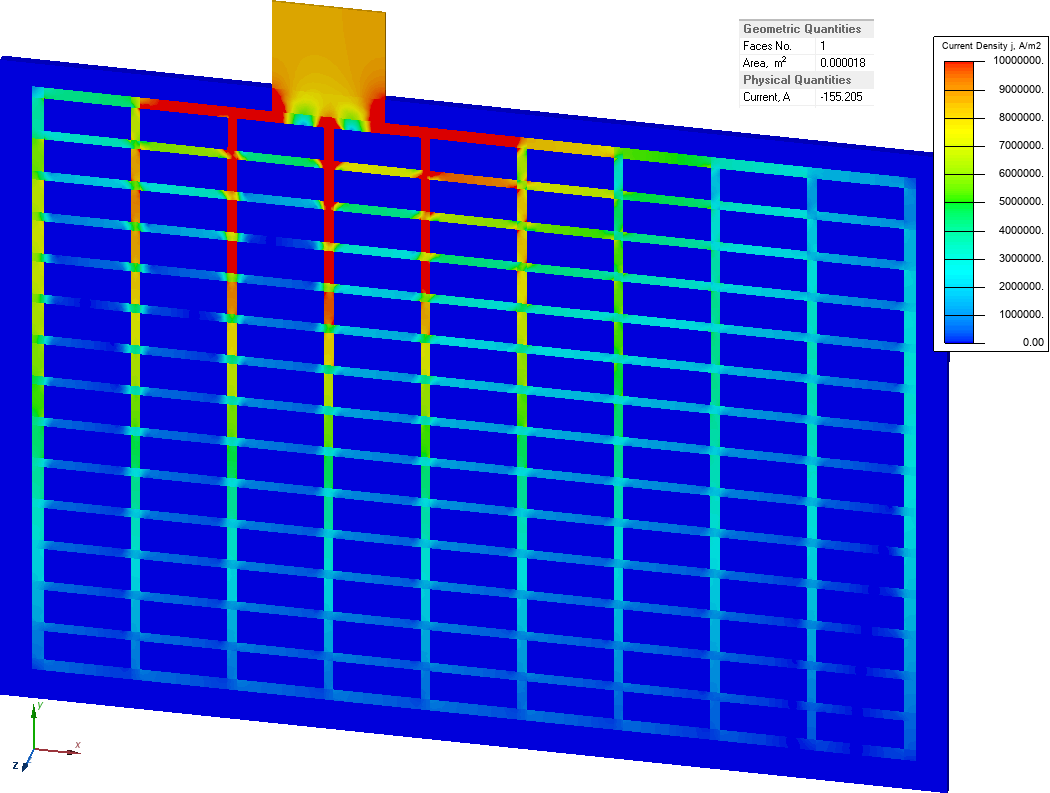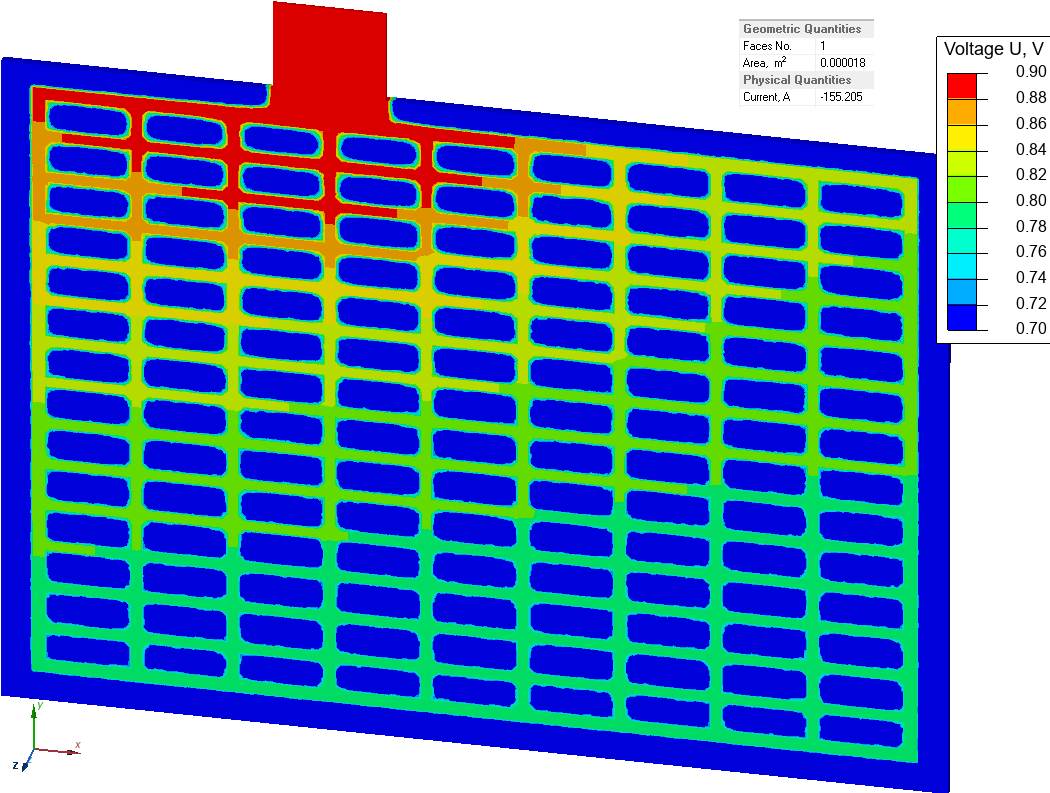
Current density and electric potential distribution in the grid electrode
Engineering question
Set up a 3D QuickField DC Conduction problem for a lead-acid battery and evaluate internal resistance from computed field results.
lead-acid battery cells, battery grid electrodes, electrochemical storage cells
Download
Simulation problem
Problem Type
3D problem of DC conduction.
Electric conductivity of lead 4.8 MS/m;
Electric conductivity of electrolyte 30 S/m;
Calculate the resistance of the cell.
To calculate the resistance we can apply a potential difference ΔV and measure the electric current value I. The resistance of a cell is then R = ΔV / I
We choose V+ = 1 V and V- = -1 V. So the potential difference is
ΔV = (V+) - (V-) = 2 V.
The model features symmetry, so only a positive grid-electrode is shown. On the plane of symmetry the U = 0V boundary condition is set.
Electric current value is 155 A. Resistance of the cell is R = 2/155 = 12.9 mOhm.

Related examples